
Pharma Services
Successful drugs of the future will be built on a bedrock of strong data
We distill complex biology into actionable data for ongoing drug development. With our dynamic combination of whole genome sequencing (WGS) and in-house bioinformatics tools and expertise, we take a holistic approach to patient stratification and biomarker analyses.
Over the past 20 years, FDA-approved drugs using pharmacogenomic (PGx) information have risen from about 10% to above 30% – an increase of over 200%1. And yet, many novel therapeutics fail in Phase 3 trials, with an estimated 57% stopped due to a lack of efficacy and another 17% due to patient safety concerns2. But these drugs may be safe and effective in the right group of people – those with a certain genomic profile.
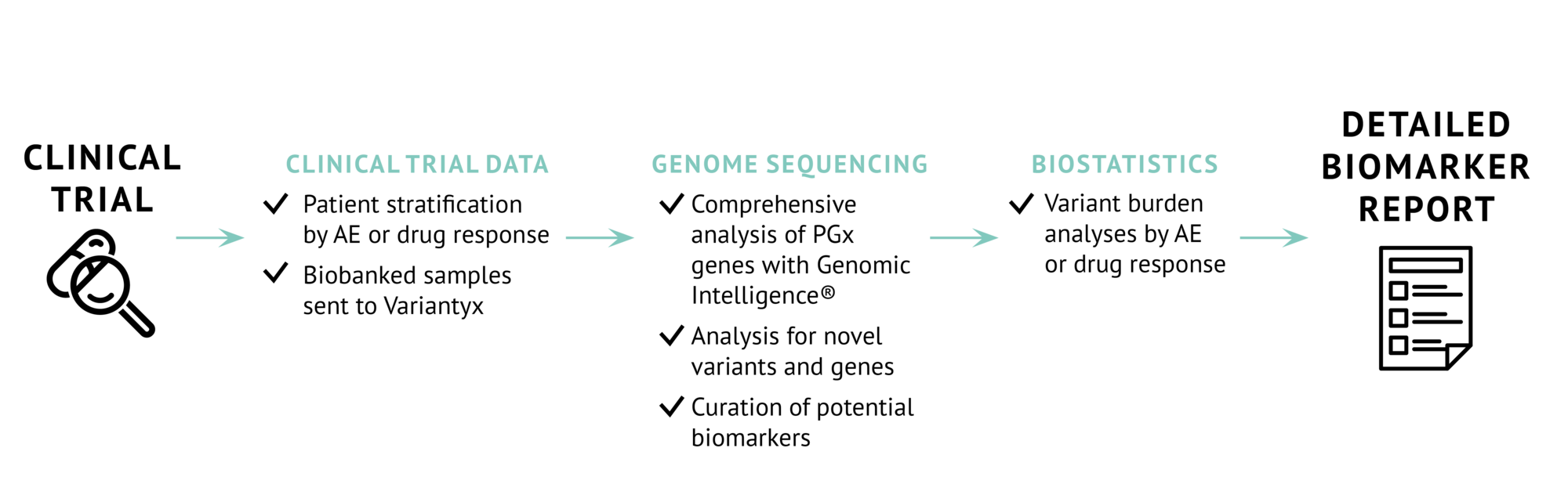
Biomarker analyses and exploration with WGS
Starting analysis with WGS allows for research-based analyses that go beyond traditional pharmacogenomics. While whole exome sequencing and genotyping arrays can identify known PGx variants, they cannot accurately identify or characterize important genomic variants. Our WGS method and analysis is a single-assay method to detect all manner of potentially important variants: intronic variants, non-coding variants, structural and copy number variants, as well as chromosomal rearrangements.
We take an agnostic approach to biomarker identification from a whole genome data by focusing on three broad areas for analysis:
This hypothesis-free design leads to maximizing biomarker discovery for patient stratification.
A cost-effective, high-value insurance policy for expensive clinical trials
Combining clinical trials data with participant genomic profiles leads to mining valuable data and may identify predictive genomic biomarkers. Using deep-phenotyping with genomic data combined with multiple statistical approaches can lead to robust patient stratification for drug safety and effectiveness and new life to a molecule or biologic. Partnering with Variantyx before clinical trials begin will help lay a foundation to collect the necessary samples and data to analyze efficacy or safety issues.
An oncology biopharma partner case study
We are currently working with a well-known oncology biopharma partner on biomarker analysis from phase II trial data. After a significant subset of patients experienced different adverse effects, they sought our expertise to stratify these patients.
A subset of the biobanked patient samples were divided into two cohorts – one that experienced adverse effects while the other that did not. Following WGS, the cohorts were analyzed for potential genetic biomarkers using multiple analysis techniques. We identified multiple significant biomarkers, leading to promising findings and continued analysis with additional patient data.
Our combination of methods, technology, and data enables precision medicine through better biomarker discovery and companion diagnostic development that may ultimately save a molecule or biologic, providing a smoother path to FDA approval.